
The plot above can help explain a thyroid lab test : high TSH, low T4 are symptoms of hypothyroidism and low TSH, high T4 are symptoms of hyperthyroidism.
In early stage of thyroid problems, high TSH and normal T4 are signs of subclinical hypothyroidism while low TSH and normal T4 indicate subclinical hyperthyroidism.
Thyroid disease is a global health problem that impacts a significant population—including those in developed economies—despite significant awareness, widespread testing and low cost of treatment.
Why? Because thyroid glad disorders are common even in young people, and more so among women (especially during pregnancy).
Another reason is too much or too little iodine in our diets. Iodine is very important for a healthy thyroid: almost 75% of the iodine in our body is used by this gland alone.
But even in iodine-sufficient areas, such as US and Europe, thyroid dysfunction is fairly common due to autoimmune diseases. Sometimes the problem might be too much of it.
That's why, iodine from supplements or through fortified foods are hot issues.
For decades, salt was fortified with iodine across the globe. However, frequent resurgence of thyroid diseases in many previously iodine-sufficient regions is a constant problem. Therefore, the constant need for monitoring and testing is important for public health.

Thyroid is a relatively small, butterfly-shaped gland, that sits in our throat.
It is one of the most important glands for our well-being because it plays many critical roles, e.g., in maintaining body temperature, mental health, and a baby's development during pregnancy.
Thyroxine (T4) and triiodothyronine (T3) are released by thyroid and are controlled by optimal (TSH) levels (thyroid-stimulating-hormone). TSH is released by the pituitary–another small glad in the brain.
The key role of thyroid hormones is in controlling metabolism. They are also important for neurological development of newborns, normal growth in childhood, and for reproduction.

Thyroid and Pregnancy – how thyroid impacts pregnancy.
Normal TSH Levels – what are healthy levels and why.
Thyroid and Iodine – understand iodine’s role in thyroid health.
A TSH Blood Test for Low Thyroid Hormones – measures TSH, free T4, free T3 levels for early detection of problems.
The Difference Between Hypothyroidism and Hyperthyroidism – a quick look at key differences.
Across the world, we seem to understand the role of thyroid hormones in every day health. But, despite such high awareness, thyroid disease is a global health problem, particularly during pregnancy and childhood.
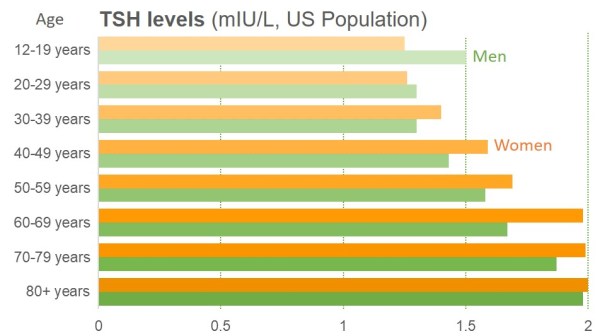
High TSH levels over 4.5 mIU/L or low TSH below 0.1 mIU/L indicate beginning of thyroid problems. The charts below suggest they are common even in young people.
In US alone, approximately 6 in 100 people (or over 18 million total) had some form of mild or serious thyroid disorder according to a 30 year old survey (NHANES III 2002). The total is certainly a lot more today.
About 80% of these cases are due to low thyroid hormones, or hypothyroidism. The remaining cases are due to hyperthyroidism, a condition of high thyroid hormones.
Normally, high TSH levels with normal T3 and T4 are first signs of the problems. Our risk of hypothyroidism is highest under 20-years and over 60-years of age.
4.5 mIU/L is the lower limit for hypothyroidism in a thyroid lab test
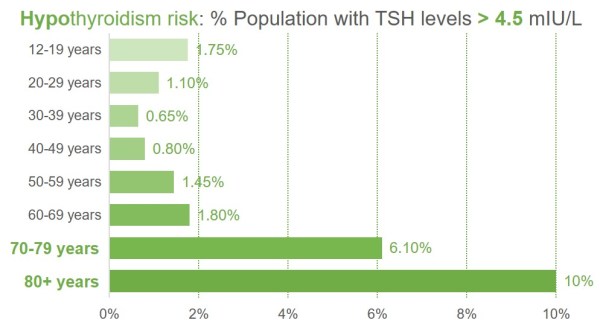
Source: National Health and Nutrition Examination Survey (NHANES III): Serum TSH, T4, and Thyroid Antibodies in the United States Population (1988 to 1994) in The Journal of Clinical Endocrinology & Metabolism, Volume 87, Issue 2, 1 February 2002, Page 489.
The risk of hyperthyroidism is highest for seniors. But surprisingly, underactive thyroid symptoms in young women are also high. This can affect their ability to get pregnant and have healthy babies.
This plot shows low TSH levels by age (with approximate cut-off taken here at 0.4 mIU/L, to include borderline hyperthyroid cases).
0.1 mIU/L is the approximate upper limit for hyperthyroidism in a thyroid lab test
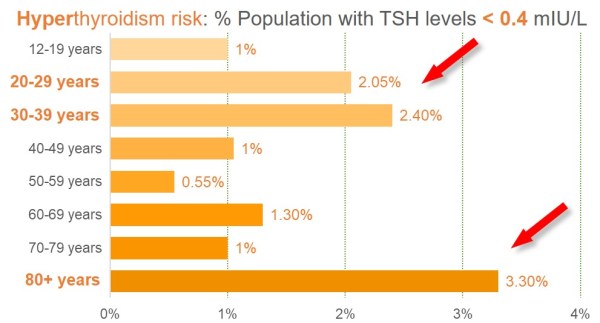
Source: National Health and Nutrition Examination Survey (NHANES III): Serum TSH, T4, and Thyroid Antibodies in the United States Population (1988 to 1994) in The Journal of Clinical Endocrinology & Metabolism, Volume 87, Issue 2, 1 February 2002, Page 489.
The hypothalamus-pituitary-thyroid (HPT) axis controls thyroid function.
First, hypothalamus senses the hormones produced by thyroid gland and releases TRH (thyrotropin-releasing hormone). Then, pituitary detects the TRH levels and releases TSH (thyroid stimulating hormone).
Next, based on TSH levels, thyroid releases two hormones: thyroxine (also called T4 due to four iodine atoms in it), and triiodothyronine (also called T3, with three iodine atoms). T3 is a simple conversion from thyroxine after loss of one iodine atom. Finally, hypothalamus senses T4, T3 to tweak TRH levels and the closed-loop cycle continues.
Any issues with this closed loop result in thyroid problems.
When one of the glands in this loop can't produce sufficient hormones, others compensate. For example, when thyroid can't make sufficient T4, pituitary increases TSH levels to drive it harder to release more T4. This continues until thyroid fails & more TSCH can't compensate anymore.
That's the main reason why testing for TSH alone might not be sufficient. A lab test should always look at multiple markers to understand if all of components of the HPT axis are functioning at their optimum.
The graph below highlights this point: As we move left along the top arrow, pituitary raises TSH levels as T4 declines. Here, the underactive thyroid with low T4 and high TSH leads to mild or subclinical hypothyroidism symptoms at first. But, then, as thyroid gland completely fails, even higher TSH levels can't increase T4, which leads to full blown symptoms of severe or clinical hypothyroidism.
Similarly, on the right side of this graph when overactive thyroid gland produces too much T4, the pituitary slows down to reduce TSH until they hit almost zero. This early stage of high T4 and low TSH is symptom of subclinical hyperthyroidism. Then, continuous overproduction of T4 causes symptoms of overactive thyroid gland.
Thyroid lab ranges from a 2002 Health survey of US population:
Mild hypothyroidism: TSH over 4.5 mIU/L (or 4.5 uIU/mL)
Severe hypothyroidism: TSH over 4.5 mIU/L and T4 below 4.5 ng/dL (or 58 nMol/L)
Mild hyperthyroidism: TSH below 0.1 mIU/L (or 0.1 uIU/mL)
Severe hyperthyroidism: TSH below 0.1 mIU/L and T4 above 13.2 ng/dL (or 170 nMol/L)
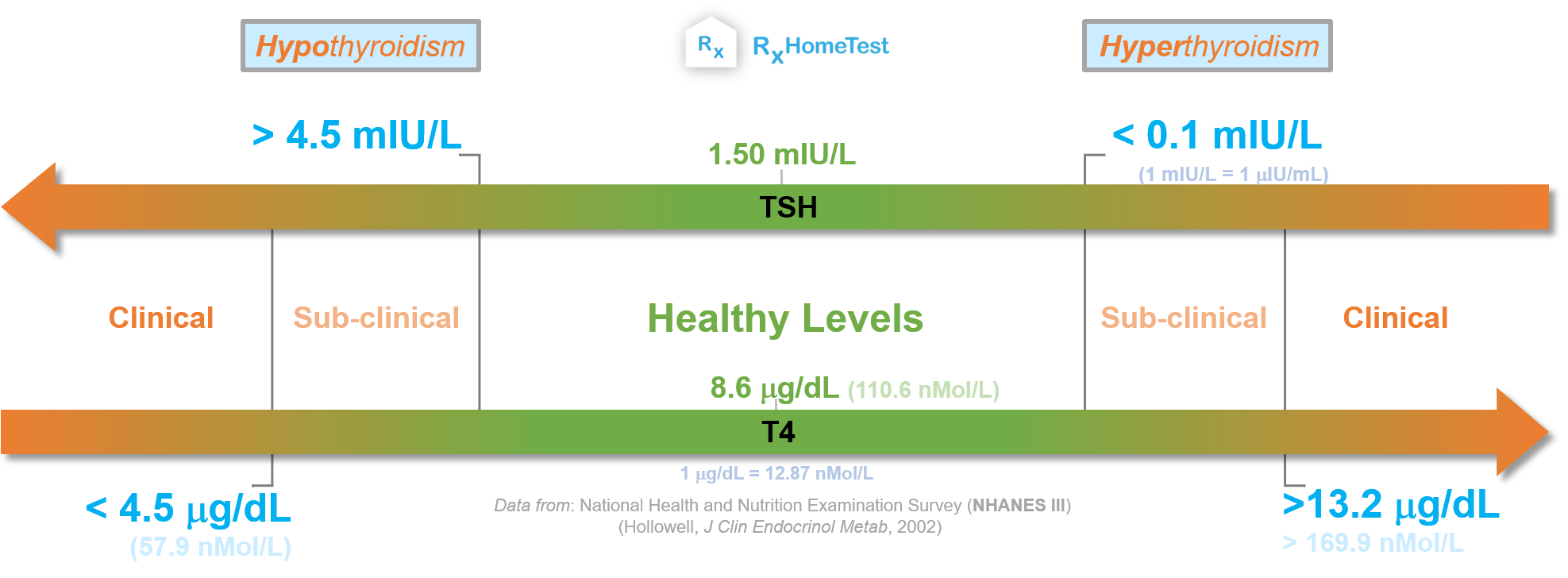
Hypothyroidism (an underactive thyroid) and hyperthyroidism (an overactive thyroid) commonly arise from pathological processes (e.g., swelling or tumor) in the thyroid gland (therefore, called primary thyroid disease). In rare cases, they can arise from disorders of the hypothalamus or pituitary (secondary or central hypothyroidism). Peripheral causes such as ovarian tumor (struma ovarii), or thyroid cancer may also appear.
Since TSH controls T4 and T3 release, diagnosis of thyroid problems starts with checking TSH levels.
When TSH levels rise above 4.5 mIU/L (milli-international-unit-per-liter) they reach the clinical definition of hypothyroidism. In contrast, too much T4 will slow down TSH release, and levels below 0.1 mIU/L result in hyperthyroidism. One should pay close attention to whether T4 or free-T4 is measured (free-T4 is active form & bound T4 is not measured).
The abnormally low or high TSH levels are first signs of a potential thyroid problem. As long as the T4 levels are within healthy range, the conditions remain subclinical and do not result in a full blown thyroid problem.
What if it’s not possible to keep up with abnormal TSH levels, and underproduction or overproduction continues? That’s clinical or overt stage of thyroid disease with potentially serious consequences.
T4 levels below 57.9 nMol/L (nano-moles-per-liter) or 4.5 ng/dL (nano-gram-per-deci-liter) combined with high TSH (above 4.5 mIU/L) is clinical or overt hypothyroidism. On the other hand, levels above 169.9 nMol/L or 13.2 ng/dL in combination with low TSH (below 0.1 mIU/L) is clinical hyperthyroidism. Both have serious health impact. See this reference for unit conversion.
Both low and high levels of iodine impact health negatively.
Alas, two-third of the world is naturally iodine-deficient. That’s why almost 120 countries mandate iodine fortification of salt in diets to ensure an adequate supply. In general, coastal areas with plenty of sea food have high iodine supply.
As of 2016, 110 countries have sufficient iodine supply in their diets and 19 countries have persistently insufficient iodine in diets, according to a 2016 report by the Iodine Global Network, a non-profit advocacy group. Clearly, iodine fortification of all food-grade salt has a long way to go (Dasgupta 2008).
A growing number of countries—ten as of 2016—have too much iodine in their diets.
In some parts of the world, fortification or jump in iodine intake has seen increases in cases of toxic nodular goiters. Cases jump post-iodization, causing fatalities as a result of cardiovascular complications.
People on thyroid hormone replacement medicines often remain hypothyroid or sometimes become hyperthyroid which is why it's important to monitor daily dose in our diets.
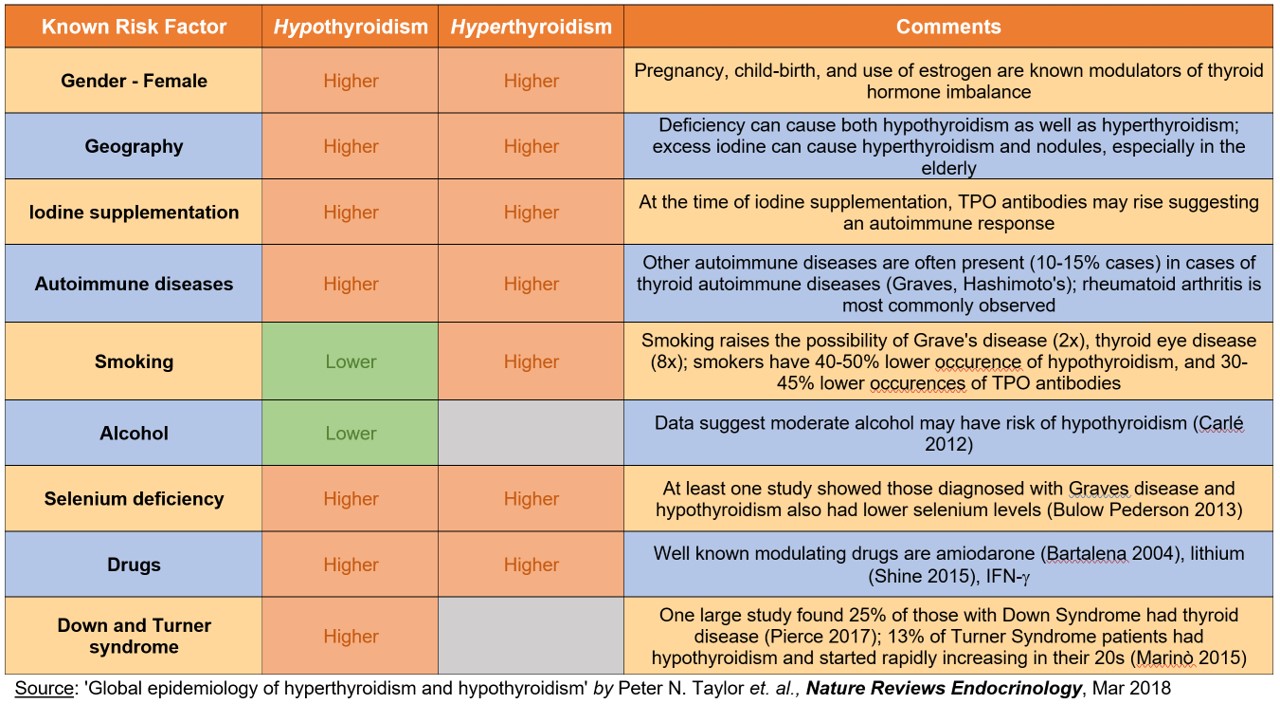
The list of risk factors is long, as the following table shows (Taylor 2018).
Everybody is at some risk (almost): as the prevalence in general population is reasonably high at 3.7% for hypothyroidism (TSH > 4.5 mIU/L) and 0.5% for hyperthyroidism (TSH less than 0.1 mIU/L).
Gender: Women are at much higher risk than men, and pregnancy causes fluctuations in thyroid levels. Women of reproductive age (12-49 years) are at much higher risk of thyroid problems.
Age: those 80 years or older have 5-times higher odds than those at 12-49 years.
Race: African-Americans tends to have lower TSH levels and higher rates of hyperthyroidism than Hispanic and White populations.
Geography: one-third of the world is iodine deficient with high rates of hypothyroidism. But coastal regions are more iodine-rich causing higher rates of hyperthyroidism.
Medication or supplements: certain medicines, e.g. levothyroxine, lithium, and amiodarone are known modulators of thyroid levels. Sudden iodine supplementation can also negatively affect thyroid health.
Here's the full list of known risk factors of thyroid dysfunction:
Even if you are doing everything right, thyroid disorder can affect anyone.
High TSH and low thyroid hormones are observed in approximately 3.7% of US population, based on data from 1999-2002. At this rate, it corresponds to 12 million people today with some form of potential hypothyroidism problem (Aoki 2008).
Low TSH and high thyroid hormones are observed in about 0.5% of US population (again, 1999-2002 data). That corresponds to 1.5M people today, with potential hyperthyroidism problem.
In regions with sufficient iodine in diet, thyroid autoimmunity is the most common dysfunction. This is confirmed through the presence of thyroid-specific autoreactive antibodies. The two well known dysfunctions are Graves's disease (an overactive thyroid when immune system attacks the gland), Hashimoto thyroiditis (an underactive thyroid when immune system attacks the gland), and postpartum thyroiditis (an abnormal thyroid hormone levels after pregnancy).
The presence of nodules in the thyroid can cause inflammation in the gland or thyroiditis (the high and low fluctuations of T4, T3 as shown in chart above).
Those taking certain medications can have adverse effects, e.g., from amiodarone and lithium.
Thyroid treatment itself can sometimes cause both hypothyroidism and hyperthyroidism.
GRAVES’S DISEASE:
It's a form of hyperthyroidism with an overactive gland such that the immune system attacks the gland itself.
Key symptoms of Graves's disease include
TED or thyroid eye disease (in 20-30% cases), with puffy, red, swollen eyes.
a much diffused and swollen goiter.
in rare cases, localized thickening of skin and thyroid acropachy that manifests as swollen hands and clubbed fingers.
Thyroid dysfunction has a gender asymmetry with women at much higher risk, and rate of dysfunction increases with age.
Data suggest hypothyroidism is approximately ten times more common in women than men (Vanderpump, 2011).
Women also tend to have higher rate of mild or subclinical hyperthyroidism (Taylor 2018).
In US, women of reproductive age (12-49 years) have a 3.1% rate of hypothyroidism.
Graves's disease (an overactive thyroid when immune system attacks the gland) is known to predominantly affect women with an 8-fold rate compared to men. The highest impact is in the age group of 30s-to-50s (De Leo, 2018).
Solitary toxic thyroid nodules, the relatively benign lesions, are more common in women than in men, and some studies have reported a 5-times higher occurrence.
Thyrotoxicosis, i.e., excessive thyroid hormones, from the iodine-rich compound amiodarone is more common in iodine-deficient areas but have a 3x higher prevalence among men.
Pregnancy is a period of large fluctuations in thyroid hormones.
In pregnant women, an increase in demand and relatively higher urinary excretion result in iodine deficiency.
Thyrotoxicosis or occurrence of excess thyroid hormone, is significantly high during pregnancy. Estimates of incidence are 0.2% for overt or clinical thyrotoxicosis and 2.5% for subclinical thyrotoxicosis (Cooper 2013; Korevaar 2017).
In US, studies estimate the rate of hyperthyroidism is 6 per 1000 pregnant women per year (Korelitz 2013).
The greatest risk of hyperthyroidism is pregnant women seems to be in the first trimester.
Graves's disease is the most common cause of thyrotoxicosis in pregnancy, although other causes, such as toxic nodules and goiters, are known to occur during pregnancy.
Subclinical hypothyroidism before 20 weeks of pregnancy is associated with an increased risk of miscarriage (Zhang 2017).
However, occurrence of hyperthyroidism during pregnancy might be overestimated, mostly due to inclusion of cases of gestational thyrotoxicosis—a benign and transient disorder of pregnancy that typically occurs in the first trimester.
Thyrotoxicosis management in pregnancy is highly complex. It is necessary to address the risk of mother’s hyperthyroidism without fetal harm from transfer of maternal antibodies through placenta and thionamide drugs.
Globally, in iodine-sufficient areas of developed countries, the prevalence of hypothyroidism during pregnancy is approximately 2% (Korevaar 2017; Medici 2015).
Isolated hypothyroxinaemia (which is defined as free T4 in lowest 2.5% of population but with normal TSH) is associated with adverse pregnancy results, including premature birth (Korevaar 2013).
Congenital hypothyroidism—the thyroid deficiency in newborns—nearly doubled in US in a 15-year period from 1987 at 1 in 3,985 to 1 in 2,273 in 2002. This is in part due to increased ethnic diversity and lowering of TSH cut-off limit.
Thyroid risk increases with age but those in 20-to-40 year age group are at surprisingly higher risk for hyperthyroidism.
Those 80 years or older have 5-times higher odds than those 12-49 years (Hollowell 2002).
Seniors are more susceptible to thyroid nodules and hyperthyroidism, especially in iodine-deficient regions.
Graves's disease–an overactive thyroid when immune system attacks the gland–is more common in younger population. They also tend to have higher thyroid hormone levels, with more likelihood of overt or clinical hyperthyroidism than mild or subclinical hyperthyroidism.
Graves disease has the highest impact in age group of 30s-to-50s and predominantly affects women with an 8-fold impact compared to men (De Leo 2016).
Surprisingly, mild or subclinical hyperthyroidism is more common in two age groups: 20-40 years old and those 80+ years old (NHANES III, Hollowell 2002). That’s why many people experience thyroid problem in youth.
Older population—with well known nodular goiter in areas of chronic iodine deficiency and trying iodine supplements—is at higher risk of iodine-induced hyperthyroid. The condition is also known as the Jod–Basedow phenomenon.
Elderly patients with potential cardiac disease and limited access to health care are potentially at the risk of iodine-induced hyperthyroid.
In elderly patients, toxic nodular goiter is the most frequent cause of thyrotoxicosis—a condition with too much thyroid hormones.
Data suggests that in iodine-sufficient countries, the prevalence of hypothyroidism ranges from 1% to 2% , rising to 7% in individuals aged between 85 and 89 years (Gussekloo 2004).

A significant variation in TSH levels is common across various ethnic groups.
Ethnicity has strong correlation to subclinical hyperthyroidism with highest prevalence at 0.4% among African-Americans, 0.3% in Mexican Americans, and 0.1% in white Americans (Hollowell, 2002). In Asia, subclinical hyperthyroidism ranges between 0.43% to 3.9% of the population.
Similar trend exists for people of mixed races: compared to non-Hispanic whites, non-Hispanic African-Americans had a 54% lower risk of hypothyroidism and 3.2-times higher risk of hyperthyroidism. Mexican Americans had the same risk as non-Hispanic whites for hypothyroidism, but a 2-fold higher risk for hyperthyroidism (Aoki 2008). Data from Brazil show same pattern: black individuals have the lowest prevalence of hypothyroidism and those of dual heritage and white individuals have a higher prevalence.
Ethnicity seems to influence the risk of developing certain thyroid disease complications. For example, risk of Graves's ophthalmopathy (or TED, thyroid eye disease) is six times more common in white populations than in Asian populations (Tellez and Cooper 1992).
The rare but serious complication of hyperthyroidism called thyrotoxic periodic paralysis is much more common among Asian men. Studies from China and Japan (Okinaka 1957) show periodic paralysis occurs in 2% of population compared to 0.2% in North America (Kelley 1989). Genetic studies show variations in certain HLA haplotypes, such as DRw8, A2, Bw22, Aw19 and B17, have been identified in patients of Chinese or Japanese origin (Tamai 1987).

Thyroid dysfunction correlates to iodine intake which strongly depends on geography.
Almost one-third of world population lives in iodine-deficient areas. Some live with severe iodine deficiency causing devastating consequences of neurological development to fetuses and children.
Thyroid nodules are more prevalent in regions with iodine deficiency. On the other hand, Graves's disease (an overactive thyroid when immune system attacks the gland), Hashimoto's thyroiditis (an underactive thyroid when immune system attacks the gland) occur in regions with adequate or excess iodine.
Nodular thyroid disorders are more prevalent in areas where iodine deficiency is more common. But autoimmune thyroid disorders, including Hashimoto's thyroiditis and Graves's disease, occur more frequently in iodine-rich populations.
Higher rates of hyperthyroidism occur in iodine-deficient regions, mainly due to thyroid nodules in elderly patients.
In iodine-sufficient regions, Graves's disease can account for 70–80% of cases with hyperthyroidism.
In iodine deficient areas, Graves's disease constitutes almost 50% of all cases of hyperthyroidism, while remaining half are due to nodular thyroid disease. As an example, Graves's disease has higher prevalence in Iceland, an iodine-rich region, in comparison to a predominance of toxic multinodular goiter in Denmark that has a lower iodine intake.
Thyrotoxicosis from the iodine-rich compound amiodarone is more common in iodine-deficient areas as more patients supplement iodine using this compound.
Regions with low iodine in diet have up to 12-times higher occurrence of toxic nodular goiter compared to regions with higher iodine. They also have up to two-fold higher incidence of solitary toxic nodules–relatively benign lesion with potential risks in rare cases.
Levothyroxine prescription has been on rise, often as part of fertility treatment and other reproduction issues.
Certain medications and lifestyles play important roles in thyroid health.
Thyroid problem can arise more frequently in those taking thyroid-related medications. These include levothyroxine, estrogen, androgen, amiodarone. Additionally, lithium, monoclonal antibodies, sodium valproate (anti-epileptic), tyrosine kinase inhibitors and immune checkpoint inhibitors can cause hypothyroidism.
Tyrosine kinase inhibitors are drugs that often cause hypothyroidism. The immune checkpoint inhibitors given to advanced cancer patients are well known to cause hypothyroidism. Few of these drugs are: ipilimumab, nivolumab, pembrolizumab, atezolizumab and durvalumab.
Among those taking levothyroxine or desiccated thyroid, the adjusted risk for hypothyroidism was 4-times and hyperthyroidism was 11.4-times (Aoki 2008).
Borderline or mild cases of thyroid disorders have risen in past few decades due to increase in lower cost sensitive testing. The treatment threshold are also becoming progressively lower.
Genetic mutations into specific tissues can also cause hypothyroidism, e.g., THRα, THRβ and MCT8 (also known as SLC16A2).
Radiographic contrast agents used during X-ray imaging and CT scan can also cause iodine-induced hyperthyroidism.
Smoking status, alcohol consumption, presence of other autoimmune conditions, syndromic conditions and exposure to some therapeutic drugs also influence thyroid disease in the population.
Significant awareness and low cost high sensitivity testing has brought more awareness.
A common question to ask is: why am I always cold? Or hot? Because thyroid hormones control body temperature, this will be first signs of thyroid dysfunction.
A first step is to check TSH levels. However, checking for T4 and TPO antibodies should be included in the test for full diagnosis.
Not all testing is equal. Or, testing may not always be necessary despite an increase in awareness, easy access and falling cost. A 2017 trial of thyroid hormone therapy for elderly patients with subclinical hypothyroidism (the TRUST trial, Stott 2017) highlights how testing might be wasteful. They found that up to 60% of potentially eligible elderly individuals with an elevated TSH had returned back to normal euthyroidism and healthy hormone levels when reassessed for the trial.
Screening during pregnancy is fairly common although large fluctuations in thyroid levels during pregnancy require careful assessment of the timing.
Up to 50% cases of mild or subclinical hyperthyroidism is now attributed to the rise in levothyroxine treatment because treatment threshold has fallen over past two decades (Taylor 2014, Eligar 2016).
Since TSH is only one part of the HPT axis, full cycle of thyroid hormone circulation can not be completely understood with TSH alone. It is a first step to identify potential issues. But to understand the subclinical or mild form of hypothyroidism or hyperthyroidism, testing for T4 levels is necessary. Similarly, an immune attack on thyroid glands can be proactively assessed by checking the TPO antibody levels.

Order an at-home thyroid test kit.
Global epidemiology of hyperthyroidism and hypothyroidism by Taylor et. al. in Nature Reviews Endocrinology, 2018.
Serum TSH, T4, and Thyroid Antibodies in the United States Population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III) by Hollowell et. al. in Journal of Clinical Endocrinology & Metabolism, 2002.
Iodine Nutrition: Iodine Content of Iodized Salt in the United States by Dasgupta, Liu & Dyke in Environ. Sci. Technol. 2008.
Serum TSH and Total T4 in the United States Population and Their Association With Participant Characteristics: National Health and Nutrition Examination Survey (NHANES 1999–2002) by Aoki et. al. in Thyroid, 2008.
The epidemiology of thyroid disease by Vanderpump in British Medical Bulletin, 2011.
Hyperthyroidism in pregnancy by Cooper and Laurberg in Lancet: Diabetes & Endocrinology, 2013.
Thyroid disease in pregnancy: new insights in diagnosis and clinical management by Korevaar et. al. in Nature Reviews Endocrinology, 2017.
Thyroid Function in Pregnancy: What is Normal? by Medici et al. Clin. Chem., 2015.
Thyroid Status, Disability and Cognitive Function, and Survival in Old Age by Gussekloo et. al. in Journal of American Medical Association, 2004.
Thyroid Hormone Therapy for Older Adults with Subclinical Hypothyroidism by Stott et. al. in New England Journal of Medicine, 2017.
Falling Threshold for Treatment of Borderline Elevated Thyrotropin Levels—Balancing Benefits and Risks: Evidence From a Large Community-Based Study by Taylor et. al. in JAMA Internal Medicine, 2014.
For monitoring latest research and literature on thyroid, visit: www.thyroid.org
Learn about hypothyroidism, hyperthyroidism, and their symptoms at All About Thyroid