
Thyroid in pregnancy is necessary for baby’s development, mother’s health, and normal pregnancy.
It is especially important for development of an infant’s central nervous system. Because the baby’s thyroid does not fully develop until second trimester, a mother’s thyroid requirements increase during pregnancy.
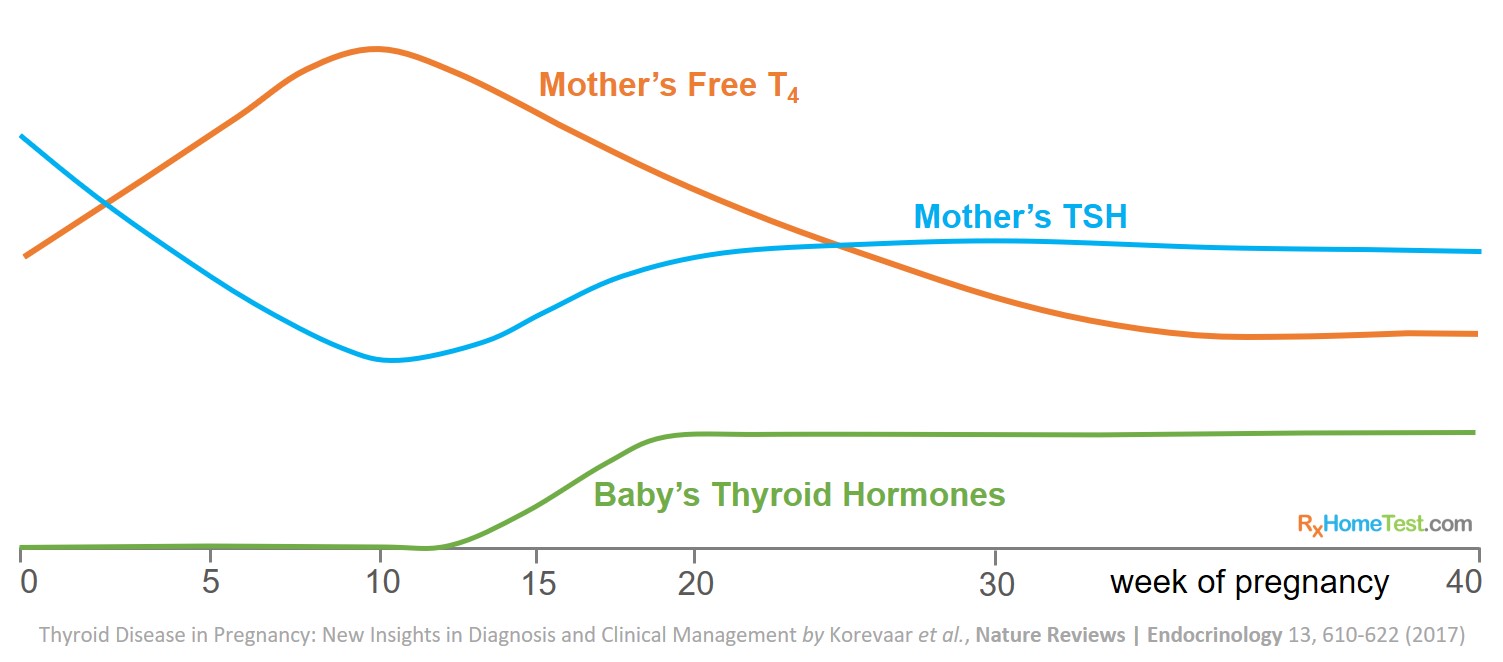
The levels fluctuate significantly in the first half of pregnancy when the baby is entirely dependent on mother’s thyroid hormones. Therefore, the role of thyroid in pregnancy is very complex.
Thyroid disease in pregnancy: new insights in diagnosis and clinical management by Korevaar et. al. in Nature Reviews Endocrinology, 2017.
The most common questions are: (1) what should my thyroid levels be to get pregnant? (2) how do thyroid levels change during pregnancy?
This article attempts to answer these question based on the available scientific research on thyroid in pregnancy.

Order an At-Home Thyroid Test.
What to Know About Thyroid Hormones in Pregnancy – thyroid hormones fluctuate every trimester and need careful monitoring.
Role of Thyroid in Your Health – how thyroid impacts everyone.
Normal TSH Levels – TSH is the first hormone tested in a lab report, what should be your levels?
At Home Thyroid Test – a test that measures TSH, free T4, free T3, and TPO.
5 Signs You Should Take an At-Home Thyroid Test – key symptoms when thyroid problems might be happening.
The Difference Between Hypothyroidism and Hyperthyroidism – symptoms tend to be opposite for low and high thyroid levels.
A Thyroid Summary in Pictures – for those who prefer short and succinct summaries.
Tips for Understanding Thyroid Test Results – ranges for TSH, free T4, free T3, and TPO.
Thyroid plays an extremely important role during pregnancy:
A baby’s thyroid does not fully develop until weeks 18–20 of pregnancy and the T4 hormone supply comes from the mother.
Mother’s free thyroxine (fT4) concentrations increase and TSH concentrations decrease from approximately eighth week through the first half of pregnancy.
In first trimester, the high concentrations of pregnancy hormone, hCG (human chorionic gonadotropin), increase thyroid hormone production and suppress TSH levels.
Pregnancy requires an increase in mother’s thyroid hormone production to ensure sufficient thyroid hormone is available for the mother and baby. That’s mainly because:
The fetus relies on mother’s thyroid hormone which increases the T4 requirement.
The protein that binds T4, thyroxine-binding globulin (TBG), increases in concentrations binding even more T4.
The mother’s urinary excretion of iodine increases which leaves less iodine for thyroid hormone production.
The placenta’s enzyme (type 3 de-iodinase) deactivates some of the T4.
These changes might expose pre-existing mild thyroid dysfunction during pregnancy.
The presence of TPO (thyroid peroxidase) antibodies can significantly impact the pregnancy and child development as the antibodies indicate autoimmune disease in mother.
Maternal T4 levels seem to affect the fetal development and TSH levels seem to affect maternal health (as TSH can not pass through placenta).
TSH necessary reference intervals during first trimester are 0.1–2.5 mIU/L and during second trimester are 0.2–3.0 mIU/L (De Groot 2012).
Screening of thyroid function is not expensive because in western countries all pregnant women have a standardized blood sample test at 8–12 weeks’ gestation. Those testing positive largely benefit from a cheap, safe, and effective treatment.

Thyroid Hormones in Pregnant Women - Plotted by week of pregnancy (Männistö 2011).
The prevalence of thyroid dysfunction during pregnancy is fairly common.
About 2% – 4% of all pregnancies have some form of thyroid related issues (Krassas 2010).
Risk of thyroid problems during pregnancy vary significantly and can affect both the mother and the child.
Overt or clinical hypothyroidism (with elevated concentrations of TSH with low concentrations of free T4) occurs in about 0.2% to 0.6% of pregnant women (Medici 2015).
Based on the birth figures of 2012, if all pregnant women from the United States, the UK, or The Netherlands were screened, the conservative annual number of cases with overt hypothyroidism (TSH > 10 mIU/L) would be 25,000, 4500, and 1000, respectively (Pop 2014).
Maternal subclinical or mild hypothyroidism (elevated concentrations of TSH with normal concentrations of free T4) is more prevalent. It occurs in 3.5% to ~18% of all pregnancies (Abalovich 2007)—however the numbers vary depending on what range of TSH your doctor defines as subclinical hypothyroidism.
The combination of high TSH concentration and positivity of TPO (thyroid peroxidase) antibody seems to synergistically increase the risk of adverse pregnancy results. Data suggest occurrence of TPO antibody in roughly one-third of pregnant women who have subclinical hypothyroidism (Casey 2006).
Overt or clinical hyperthyroidism is fairly uncommon in pregnancy.
Pregnancy data collected for 4199 women (Pop 2014) showed the prevalence of overt or clinical hypothyroidism (a TSH >10 mIU/L) was just 26 (0.62%). But a surprisingly 96% had (highly) elevated TPO antibodies.
Pathological hyperthyroidism (mainly Graves disease and toxic nodules or goiters) during pregnancy is not very common. Data show a frequency of 0.4% to 1% before pregnancy and approximately 0.2% during pregnancy (Cooper 2013).
Gestational or pregnancy-related hyperthyroidism (biochemically, high levels of free T4 and low levels of TSH) is much more frequent. It is diagnosed in about 1% to 3% of pregnancies (Cooper 2013).
In published data (Casey 2006), subclinical hyperthyroidism (TSH levels below 1.75 ng/dL or 0.42 mIU/L) were found to be 1.7% in 25,765 pregnant women. 0.4% were found with higher fT4 (which was similar to non-pregnant women in same age group of around 25 years).
Also, women with subclinical hyperthyroidism were less likely to be obese than women with TSH values between the 5th and 95th percentile (+/- 2 std dev). This positive relationship between TSH values and BMI has been well studied. It is hypothesized to represent an altered energy balance due to increased thermogenesis in obese women (Iacobellis 2005).
Subclinical or mild hyperthyroidism has not been associated with any of the commonly known adverse pregnancy outcomes (Casey 2006).
T4 plays a key role in pregnancy:
More than 99% of T4 is bound to thyroid hormone-binding proteins (TBG) and not freely available.
Therefore T4 concentrations are highly dependent on changes in TBG concentrations. The T4 concentrations are more variable during early pregnancy than free T4 concentrations.
Differences in the concentrations of free T4 explain more clearly the variation in concentrations of TSH than concentrations of free T4 do (8.0% for free T4 versus 2.5% for T4). This suggests that free T4 reflects the function of the hypothalamic–pituitary–thyroid axis more accurately than T4 (Glinoer 1990).
During early pregnancy, the free T4 is associated with adverse pregnancy and child outcomes, not T4 (Glinoer 1990).
Research suggests that free T4 is a more useful index of thyroid function during early pregnancy than T4 (Korevaar 2016).
Higher BMI associates with higher TSH concentrations (Han 2015), lower free T4 concentrations, higher free T3 (Knight 2016), and a higher T3:T4 ratio.
A Finnish study suggests shift in the TSH upper limits with BMI. This suggests higher BMI is more susceptible to hypothyroidism (upper TSH limit of 2.86 mIU/L for BMI of 20-25 kg/m2 versus 3.50 mIU/L for BMI >30 kg/m2) (Männistö 2011). The prevalence of overt hypothyroidism in very obese mothers (BMI >40 kg/m2) was found to be 11.8% (Michalaki 2006).
Factors affecting thyroid health during pregnancy include genetic, dietary, environmental and cultural factors. The differences in maternal thyroid function between and within different ethnic populations during pregnancy have been well studied (Walker 2005, Benhadi 2007, La’ulu 2011).
A higher proportion of African-American women show subclinical hyperthyroidism than Hispanic women (3% versus 1.6%). African-American women have higher serum levels of the pregnancy hormone (hCG) than white or Hispanic women.
Smoking has only limited effects on mean TSH and fT4 concentrations during pregnancy. A Finnish study of pregnant women found that smokers had TSH concentrations identical to those of nonsmokers (1.02 mIU/L). On the other hand there was a small difference in fT4 concentrations (15.02 vs 15.24 pmol/L) (Männistö 2011). However, smoking can negatively affect pregnancy in other ways beyond thyroid dysfunction.

Overt maternal hypothyroidism is one of the biggest concerns during pregnancy.
It correlates to higher risk of pregnancy complications, including premature delivery, low birth weight, miscarriage and pre-eclampsia , as well as a higher risk of detrimental effects on fetal neuro-development.
A large case–control study demonstrated that children born to women with untreated hypothyroidism have a 7-point reduction in IQ compared with children whose mothers were healthy thyroid levels (Haddow 1999). These children also had delays in motor skill development, language development and attention at 7 to 9 years of age.
Surprisingly, no data suggest that women with adequately treated hypothyroidism have an increased risk of pregnancy complications compared with women with normal thyroid function (Korevaar 2017).
The transfer of mother’s T4 to the fetus is essential for optimal fetal brain development, and levothyroxine supplementation is most common treatment for overt hypothyroidism.
Similar to overt hypothyroidism, sub-clinical or mild hypothyroidism correlates with a higher risk of pregnancy complications.
These complications include pregnancy loss, placental abruption, premature delivery, pre-eclampsia and neonatal death (Sheehan 2015, Maraka 2016, Negro 2014). However, the ranges for sub-clinical hypothyroidism need careful defining as values may vary significantly between studies and various labs.
Evidence from large population studies indicates that sub-clinical hypothyroidism is not associated with any adverse neuro-behavioral results in the baby (Korevaar 2016).
Thyroid autoimmunity is a major risk factor for sub-clinical hypothyroidism (Medici 2012). Studies show approximately one-third of women with sub-clinical hypothyroidism are found positive for TPO antibodies (Casey 2006, Korevaar 2017).
Combination of sub-clinical hypothyroidism and TPO antibody positivity is associated with a higher risk of adverse pregnancy results, such as miscarriage, gestational diabetes mellitus (glucose intolerance during pregnancy) and premature delivery (Korevaar 2013, Karakosta 2012, Liu 2014, Ying 2016).
Combination of high concentrations of TSH and TPO anti-bodies increases the risk of adverse pregnancy results compared with the presence of only high TSH concentrations or TPO antibodies. Also, for women who are TPO positive are at higher risk already for TSH concentrations even in the high-normal range (in most studies for concentrations of TSH >2.5mIU/L).
One study showed that a large percentage of pregnant women with high serum thyrotropin concentrations subsequently had clinically apparent hypothyroidism. Because the symptoms associated with hypothyroidism are nonspecific, the condition can be difficult to diagnose, as reflected by the five-year median time to diagnosis in these women (Haddow 1999).
TPO (thyroid peroxidase) antibodies are a marker of thyroid autoimmunity and they are the most important risk factor for thyroid problems during pregnancy.
Research shows women who are TPO positive have higher TSH concentrations, lower free T4 concentrations, and a higher risk of thyroid dysfunction during pregnancy than women who are TPO negative (Thangaratinam 2011, Medici 2012).
Intriguingly, being TPO positive by itself correlates with an elevated risk of miscarriage and premature delivery (Korevaar 2013).
Data show that women testing positive for TPO antibodies are at high risk for thyroid-related adverse results, especially if TSH concentrations are elevated or in the higher end of the normal range (Korevaar 2017).
In one study, the presence of high serum concentrations of thyroid peroxidase (TPO) antibodies in 77 percent of the women with hypothyroidism indicates that chronic autoimmune thyroiditis was the most frequent cause of hypothyroidism (Haddow 1999).
Sub-clinical hyper-thyroidism does not associate with any adverse pregnancy results (Casey 2006).
In one large study where a total of 25,765 women underwent thyroid screening and delivered single babies. 433 (1.7%) of them had sub-clinical hyperthyroidism (below 1.75 ng/dL or 0.42 mIU/L). Pregnancies in women with sub-clinical hyperthyroidism were less likely to complicate from hypertension.
Most cases of pregnancy related sub-clinical hyperthyroidism are of short term. This along with their physiological nature makes it hard to collect enough scientific data.
Few studies have shown that higher free T4 concentrations are associated with reduced birth weight and reduced child cognitive function (Korevaar 2013, Haddow 2014).
Sub-clinical hyper-thyroidism does not associate with any adverse pregnancy results (Casey 2006).
In one large study where a total of 25,765 women underwent thyroid screening and delivered single babies. 433 (1.7%) of them had sub-clinical hyperthyroidism (below 1.75 ng/dL or 0.42 mIU/L). Pregnancies in women with sub-clinical hyperthyroidism were less likely to complicate from hypertension.
Most cases of pregnancy related sub-clinical hyperthyroidism are of short term. This along with their physiological nature makes it hard to collect enough scientific data.
Few studies have shown that higher free T4 concentrations are associated with reduced birth weight and reduced child cognitive function (Korevaar 2013, Haddow 2014).
During pregnancy, two major types of overt hyperthyroidism can occur:
A rare pathological form that mainly affects women with Graves disease. Or diseases characterized by autonomous thyroid hormone production, e.g., multinodular toxic goiter or toxic adenomas.
A form with transient elevations in thyroid function due to high pregnancy hormone (hCG) levels. These typically peak around the 10th week of pregnancy.
Pregnancy related hyperthyroidism occurs in 0.3–1.0% of all pregnant women. This is as defined by TSH <2.5th percentile and a free T4 above the 97.5th percentile or 2 std dev (Casey 2006, Korevaar 2013, Springer 2009).
In the first type, prevalence rates in western countries are 0.5-1.3% for pre-existing Graves disease, 0.05% for new-onset Graves disease, and 0.1% for autonomous thyroid hormone production (Cooper 2013, Carle 2011).
The disease often presents with clear biochemical abnormal signs. For example, as suppressed TSH concentrations with high free T4 concentrations. They are typically above 1.5x the upper limit of normal. It has clear symptoms such as palpitations, tremor or anxiety and with a high risk of negative pregnancy results.
Several studies link the pathological forms of pregnancy hyperthyroidism to a higher risk of pre-eclampsia, preterm (early) birth, low birth weight and maternal heart failure (Cooper 2013, Sheffield 2004, Sahu 2010, Luewan 2011, Millar 1994, Pillar 2010).
A large American study of 417 pregnant women diagnosed with hyperthyroidism and ~217,000 controls found those diagnosed with hyperthyroidism had 1.8x higher risk of pre-eclampsia (Männistö 2013).
Women diagnosed with hyperthyroidism also had a 1.2x higher risk of threatened preterm birth. They showed 1.8x higher risk of late preterm birth. Also, they had 3.7x higher risk of maternal admission to intensive care versus control participants. Another Danish study had similar conclusion. It showed women diagnosed with hyperthyroidism have higher risks of miscarriage, stillbirth, or preterm birth. Also, there is risk of lower birth weight and having a child with attention deficit hyperactivity disorder (Andersen 2014, Anderson 2013).
Overall, women with hyperthyroidism show a 3.9x higher risk of pre-eclampsia. They have a 2.2x higher risk of fetal growth restriction and 1.7x higher risk of preterm birth. They also face a 3.6x higher risk of induction of labor than control participants (Aggarwal 2014).
Some of these women do not have symptoms of hyperthyroidism. Others are classified as being transiently thyrotoxic and require treatment with therapeutics such as propranolol to relieve symptoms.
Thyroid malfunction during pregnancy increases risk of various adverse outcomes. Outcomes may be miscarriage, lower than normal fetal growth, hypertensive disorders, early delivery, and a decreased child IQ (Krassas 2010). Below we discuss each of these risks in detail for thyroid in pregnancy.
Mothers with sub-clinical or mild hypothyroidism are at an increased risk of pregnancy loss (Medici 2015).
Studies show women with serum TSH concentrations of 2.5–5.0 mIU/L had a 6.1% risk of pregnancy loss, compared to 3.6% in women with a TSH concentration below 2.5 mIU/L (Negro 2010).
There is a linear correlation between TSH and pregnancy loss according to a Dutch study of 2497 pregnant women. Total miscarriages, fetal and neonatal deaths increase by 80% on every doubling of the TSH concentration. However, this is small dataset with only 27 cases (Benhadi 2009).
Another analysis from UK combined 202 pregnancies of miscarriage or fetal loss and 3592 normal pregnancies. It showed pregnancies with complication by child loss had higher mean TSH and lower fT4 concentrations. And a higher prevalence of TSH concentrations (Ashoor 2010).
Early-pregnancy TSH concentrations (>95th percentile) associate with an increase in 3.66-times risk of miscarriages in an Australian pregnancy cohort (Schneuer 2012).
Study of 17,298 pregnant women shows subclinical hypothyroidism (elevated TSH and normal free T4) was associated with a slightly increased risk of premature delivery at <34 weeks (4% vs 2.5%); borderline significantly associated with prematurity <32 weeks (2.5 vs 1%), and not associated with prematurity <36 weeks (7 vs 6%) (Casey 2005).
When TPO antibodies were present, a 1.9x and 2.5x higher risk of premature delivery at <37 and <34 weeks, respectively was seen among women with a TSH >4.0 mIU/L. However, no associations were seen with TSH >2.5 mIU/L.
Studies in women presenting for prenatal care, subclinical hyperthyroidism (n=433) was not associated with prematurity <=36, <=34, and <=32 weeks (Casey 2006). The subclinical hyperthyroidism (n=224 cases) was not associated with prematurity <37 and <34 weeks either (Männistö 2009).
The main benefit—an increase of approximately 4 points in IQ scores — would occur in the children of women with serum thyrotropin concentrations at or above the 98th percentile (Haddow 1999).
A secondary benefit is saving lives of women systematically identified and treated in time.
Hypertensive disorders, including pregnancy related hypertension and (pre)eclampsia, are common during pregnancy and are an important cause of maternal and fetal morbidity and mortality (Bellamy 2007).
Both hypo- and hyperthyroidism have vascular effects, including endothelial cell dysfunction (Klein 2001), and associate with an higher risk of hypertensive disorders during pregnancy in limited studies conducted so far.
Study of the correlation between free T4 and birth weight in a population-based cohort (without any overt thyroid dysfunction) found a statistically significant negative relation between fT4 and birth weight (Shields 2011).
Study of Dutch pregnant women confirmed above result that high-normal fT4 concentrations are associated not only with lower mean birth weights, but also with more SGA (small size for gestational age or smaller babies) and lower than 2500-g newborns (Medici 2013). However, these children do not suffer from more labor/delivery complications (Haddow 2014).
Low birth weight is a risk factor for cardiovascular and psychiatric diseases in later life (Barker 2006; Gale 2004).
By definition, hypothyroxinemia in pregnancy is a normal TSH but low free T4 value. A value below the 2.5th percentile or 3-std dev lower qualifies.
In 3,659 mother–child pairs from a prospective birth cohort, maternal hypothyroxinemia (defined as a TSH <2.5 mIU/L with free T4 <5th percentile) was associated with a 1.8x higher risk of expressive language delay at both 18 & 30 months of age (Henrichs 2013).
In same study, hypothyroxinemia correlates with a 2x higher risk of nonverbal cognitive delay.
Follow-up data subsequently revealed that maternal hypothyroxinemia (free T4 <5th percentile and TSH <2.5 mIU/L) was associated with a non-verbal IQ that was 4.3 points lower in these children at 6 years of age (Ghassabian 2014). Free T4 concentrations <10th percentile were associated with a child IQ that was 1.5–3.8 points lower than that associated with maternal free T4 concentrations in the middle 80 percentiles (Korevaar 2016).
A Spanish study involving 1,643 mother–child pairs showed that a clear relationship existed between low maternal free T4 concentrations and mental score–at 14 months of age, infants whose mothers had free T4 concentrations during pregnancy <10th percentile, <5th percentile and <2.5th percentile had an IQ that was 2.4, 3.4 and 4.2 points lower, respectively, than that of children whose mothers had gestational free T4 concentrations above these cut-offs (Julvez 2013).
While sub-clinical hypothyroidism relates to various negative pregnancy results, hypo-thyroxi-nemia predominantly associates with negative neuro-behavioral outcomes in the child (Korevaar 2013).
Studies suggest that sub-clinical hypothyroidism or maternal TSH concentrations better reflect maternal thyroid status. Whereas, maternal free T4 concentrations better reflect thyroid hormone availability for the fetus. This is independent of TSH concentrations (Korevaar 2016, Henrichs 2013, Julvez 2013).
The results of studies associating hypo-thyroxi-nemia with adverse pregnancy outcomes, such as premature delivery or pre-eclampsia, are not very consistent (Korevaar 2013, Casey 2007, Männistö 2010).
Iodine is an essential component of the thyroid hormones, T4 and T3. It contributes 65% and 59% of their respective molecular weights.

For women of childbearing age, the median UI (urinary iodine) concentration remains at ~130 ug/L. Overall, the prevalence of a UI concentration <50 ug/L remains at about 15%.
The median UI concentrations for the general U.S. population 2007–2008 were 164 ug/L (95% CI 154–173) (Caldwell 2011).
The recommended amount is 150 ug/day for adults, 200 ug/day for pregnant or lactating women, and lower amounts for children (Dunn 1998).
In the combined dataset for 2005–2006 and 2007–2008 NHANES, 56.9% +/- 7.9% of pregnant women had a UI concentration <150 ug/L (although sampling of only 184 women). The assessment of 100 pregnant women from Boston, Massachusetts demonstrated that 49% of them had a UI <150ug/L (Pearce 2004).
The health risk to the fetus from maternal iodine insufficiency is greatest during the first trimester. That’s because the fetus is reliant upon mother’s iodine for thyroid hormone synthesis. A maternal median UI concentration <=50 mg/L during the first half of gestation can cause thyroid abnormalities such as increased thyroid gland size and elevated TSH and thyroglobulin concentrations in the newborn (Glinoer 1995; Pedersen 1993).
A maternal UI concentration >50 ug/L during the second trimester, an increased thyroglobulin, and a decreased free T4 index can cause thyroid abnormalities in the newborn (Kung 2007; Kung 2000).
Iodine supplementation can decrease the prevalence of postpartum goiter from iodine deficiency caused by gestation and lactation (Glinoer 1992; Antonangeli 2002).
Consequences of Iodine-deficiency-syndrome include goiter, cretinism, intellectual impairment, brain damage, mental retardation, stillbirth, congenital deformities, and increased perinatal mortality (Reference: CDC).
Iodine supplementation before pregnancy versus during pregnancy appears to decrease a woman’s risk for abnormal thyroid function tests. That means a decrease in free T4 and increase in TSH. (Moleti 2008).
Individuals who were hypothyroid at this critical period frequently have permanent mental retardation. A later administration of thyroid hormone or iodine can not correct this irreversible damage. Child survival is also threatened by iodine deficiency, and several studies show that neonatal mortality decreases, sometimes by 50% or more, when the deficiency is corrected (DeLong 1997).
The damage is greater when iodine deficiency provokes hypothyroidism during fetal or early life. That’s because thyroid hormone is necessary for proper development of the central nervous system, particularly its myelination (neuron sheath formation).
Iodine may have effects independent of the thyroid as clinical studies suggest that fibrocystic breast disease is sensitive to iodine nutrition and improves when supplemented, particularly with iodine (I2) rather than iodide (Ghent 1993).
Dairy and grain products are the primary sources of iodine in the American diet (Murray 2008). In US, salt is iodized with potassium iodide and 50%–60% of the U.S. population consuming it. Only a small percentage of its salt from table salt because ~70% of dietary salt is derived from processed food, which uses mostly non-iodized salt (Dasgupta 2008).
Thyroid disease in pregnancy: new insights in diagnosis and clinical management by Korevaar et. al. in Nature Reviews Endocrinology, 2017.
Thyroid Function and Human Reproductive Health by Krassas & Glinoer in Endocrine Reviews, 2010.
Early pregnancy reference interval of thyroid hormone concentrations in a thyroid antibody-negative pregnant population by Männistö et al., Thyroid, 2011.
Thyroid Function in Pregnancy: What is Normal? by Medici et al. Clin. Chem., 2015.
Association of maternal thyroid function during early pregnancy with offspring IQ and brain morphology in childhood: A population-based prospective cohort study by Korevaar et al. Lancet, 2016.
2017 Guidelines of the American Thyroid Association for the Diagnosis and Management of Thyroid Disease During Pregnancy and the Postpartum by Alexander et al. Thyroid, 2017.
Iodine Status of the U.S. Population, National Health and Nutrition Examination Survey, 2005–2006 and 2007–2008 by Caldwell et al. Thyroid, 2011.
Management of Thyroid Dysfunction during Pregnancy and Postpartum: An Endocrine Society Clinical Practice Guideline by De Groot et. al. in Journal of Clinical Endocrinology & Metabolism, 2012.
The Attitude Toward Hypothyroidism During Early Gestation: Time for a Change of Mind? by Pop, Broeren & Wiersinga in Thyroid, 2014.
Subclinical hypothyroidism and thyroid autoimmunity in women with infertility by Abalovich et. al. in Gynecological Endocrinology, 2007.
Thyroid Disease in Pregnancy by Casey & Leveno in Obstetrics & Gynecology, 2006.
Hyperthyroidism in pregnancy by Cooper and Laurberg in Lancet, 2013.
Relationship of thyroid function with body mass index, leptin, insulin sensitivity and adiponectin in euthyroid obese women by Iacobellis et. al. in Clinical Endocrinology, 2005.